Imagine a sudden splash of drain cleaner or a spray of industrial solvent hitting your eye. In those first few seconds, your instinct might be to panic, rub your eye, or run inside to find a towel. But when it comes to chemical eye burn first aid, those instincts can be dangerous. The difference between keeping your sight and facing permanent vision loss often comes down to what you do in the first ten seconds after exposure.
A chemical splash isn't just a surface sting; it's a race against time. Corrosive substances can penetrate the Cornea is the clear, dome-shaped surface that covers the front of the eye and deeper ocular tissues almost instantly. While it sounds scary, the solution is surprisingly simple: massive amounts of water, delivered immediately and continuously. If you can start rinsing within 10 seconds, you can cut the risk of permanent blindness by 76%.
Quick Guide: Immediate Steps for a Chemical Splash
If you or someone else has just had a chemical splash, do not waste time looking for a manual. Follow these steps immediately:
- Start rinsing now: Use the nearest source of clean water (tap, shower, or bottled water). Do not wait to find "sterile" saline; tap water is perfectly fine and the delay to find saline is more dangerous than using tap water.
- Position the head: Tilt your head back and turn toward the injured side. This prevents the chemical from washing across your face and into your unaffected eye.
- Force the eye open: This is the hardest part because it hurts. Use your fingers to physically hold your eyelids open. If you don't, the chemical stays trapped in the folds of the eyelid, continuing to burn the eye.
- Flush for 20 minutes: Do not stop just because the stinging eases. Keep the water flowing over the eye for a full 20 minutes.
- Call for help: While rinsing, have someone call emergency services or get a ride to the hospital. Do not stop the irrigation to make the call.
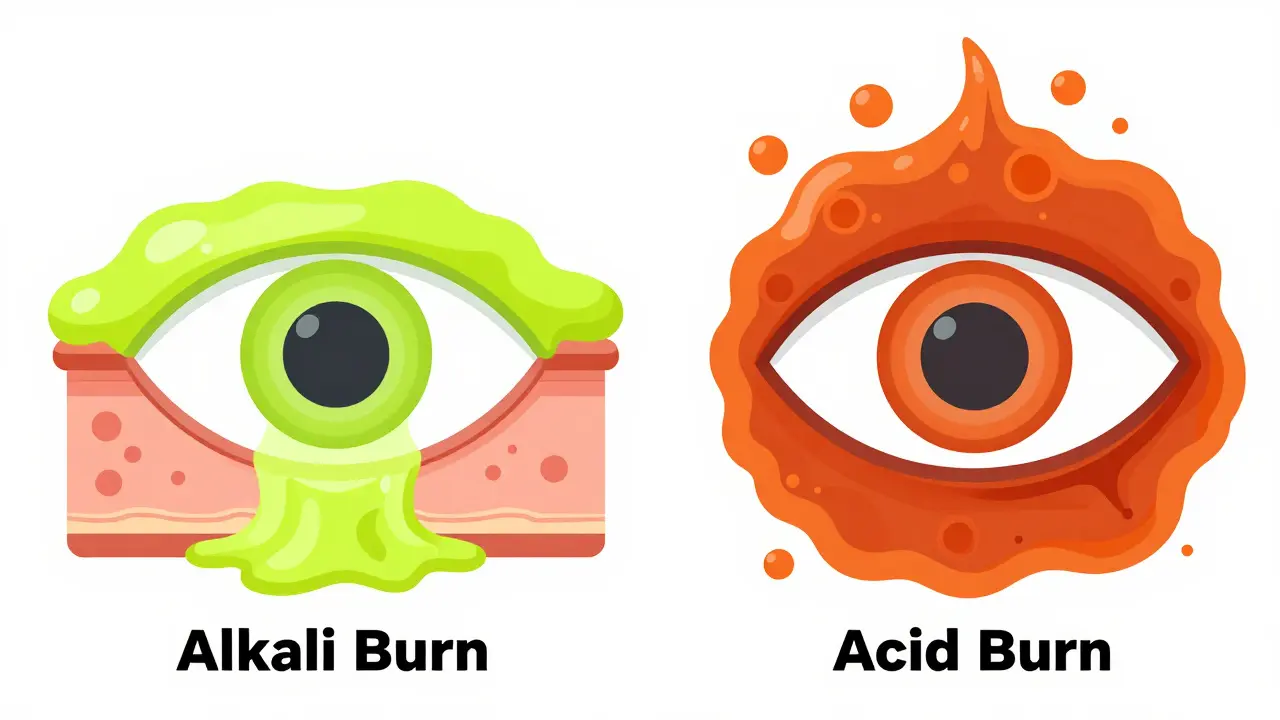
Understanding the Danger: Alkali vs. Acid Burns
Not all chemical splashes are created equal. The type of substance determines how the injury progresses. In medical terms, we look at the pH scale to understand the damage.
Alkali Burns are injuries caused by basic substances with a pH typically above 11.5, such as bleach, ammonia, or oven cleaners. These are generally more dangerous than acid burns because they cause "liquefactive necrosis," which means they melt through the eye's tissues, allowing the chemical to penetrate deeper and faster into the eye.
Acid Burns result from substances with a low pH, typically below 2.5, such as sulfuric acid or battery acid. Acids tend to cause "coagulative necrosis," which creates a barrier of damaged tissue that can actually slow down the chemical's penetration into the eye.
| Feature | Alkali Burn (Bases) | Acid Burn (Acids) |
|---|---|---|
| Common Examples | Bleach, Drain Cleaner, Lime | Battery Acid, Vinegar, Toilet Bowl Cleaner |
| Tissue Effect | Deep penetration (Melts tissue) | Surface coagulation (Creates a barrier) |
| Severity | Generally more severe/destructive | Severe, but often more localized |
| Irrigation Need | Extremely high; requires longer flushing | High; immediate flushing required |
Common Mistakes That Worsen the Injury
When panic sets in, people often do things that actually make the injury worse. Based on emergency room data, here are the most common pitfalls to avoid:
- Rubbing the eye: This is the most common mistake. Rubbing pushes the chemical deeper into the ocular tissue and can cause the crystals or corrosive agents to scratch the cornea.
- Applying pressure: Do not press on the eyeball with a cloth or your hand. You could potentially push the chemical further in or cause a corneal perforation if the tissue is already weakened.
- Using "neutralizing" agents: Never try to neutralize an acid with a base (like putting baking soda in an acid splash). The chemical reaction of neutralization creates heat, which can cause a thermal burn on top of the chemical burn. Water is the only safe universal neutralizer.
- Stopping too early: Many people stop rinsing after 5 minutes because the eye "feels better." This is a trap. The chemical may still be present in the deeper layers of the eye.
Special Considerations: Contact Lenses and Workplace Safety
If you are wearing contact lenses during a splash, they can act as a reservoir, trapping the chemical against the cornea. If the eye surface isn't too severely damaged, you should try to remove the lenses as quickly as possible during the rinsing process. However, don't spend minutes struggling with them-keep the water flowing first.
In a professional setting, the requirements are much stricter. The ANSI Z358.1 Standard is the industry benchmark for emergency eyewash stations in the workplace . If you work with chemicals, your employer must provide a station that can be reached within 10 seconds. These stations are designed to deliver a specific flow of tepid water (between 16-38°C) for at least 15 minutes. If you're in a warehouse or lab, take a moment today to actually walk to the eyewash station and ensure it works. Knowing exactly where it is can save your sight in a crisis.

Medical Treatment and Long-term Recovery
First aid is the most critical step, but it isn't the end of the process. Once you reach the emergency room, doctors will likely perform a pH test using litmus paper. They will continue to irrigate the eye until the pH returns to a neutral range (7.0 to 7.4). This process can take much longer than the initial 20 minutes of first aid.
Depending on the severity, recovery can involve various treatments. Some patients may require medicated eye drops to reduce inflammation or antibiotics to prevent infection. In the most severe cases, where the cornea is permanently scarred or perforated, a corneal transplant may be necessary. This is why the speed of that first rinse is so vital; the goal is to prevent the damage from reaching the point where surgery is the only option.
Can I use saline solution instead of tap water?
Yes, sterile saline is great, but it is not necessary. Expert ophthalmologists have stated that tap water provides no significant disadvantage compared to saline for initial emergency irrigation. The most important factor is the volume and speed of the water, not whether it is sterile. Do not waste time searching for saline if tap water is available.
How long should I actually flush my eyes?
The general rule is a minimum of 15 to 20 minutes of continuous flushing. For severe alkali burns (like lye or drain cleaner), you may need to flush for much longer, and medical professionals will continue this process until the eye's pH is neutralized.
Should I cover my eye with a bandage after flushing?
Do not apply any ointments or tight bandages unless instructed by a doctor. A loose shield may be used to protect the eye during transport to the hospital, but avoid putting pressure on the globe of the eye.
What if the chemical was just a mild household cleaner?
Even mild cleaners can cause significant irritation or chemical burns. You should still follow the 15-20 minute rinsing protocol and seek medical advice, as some chemicals can cause delayed reactions that aren't immediately obvious.
Is it okay to use a wet cloth to wipe the eye?
No. Wiping or rubbing can cause further mechanical damage to the cornea and may push the chemical deeper into the tissue. You need a continuous stream of running water to flush the chemical away from the eye entirely.
Next Steps and Troubleshooting
After the initial emergency, your path to recovery depends on the severity of the burn. For those who suffered a significant splash, follow-up appointments with an ophthalmologist are mandatory to monitor for corneal scarring or secondary infections.
If you are a business owner or manager, audit your safety equipment. Ensure your eyewash stations are not blocked by boxes or equipment and that employees are trained not just on where the station is, but how to physically hold their eyelids open during a flush. Practical, hands-on training is significantly more effective than simply handing out a safety manual.

