When someone is diagnosed with type 1 diabetes, they’re not just getting a sugar problem-they’re facing a full-body immune attack on their pancreas. This isn’t just about needing insulin. It’s about a broken immune system that mistakes insulin-producing cells for invaders and destroys them. The pancreas doesn’t just stop working. It’s under siege. And understanding that shift-from seeing T1D as a simple insulin deficiency to recognizing it as an autoimmune pancreatic disease-changes everything about how it’s managed.
What Actually Happens in the Pancreas?
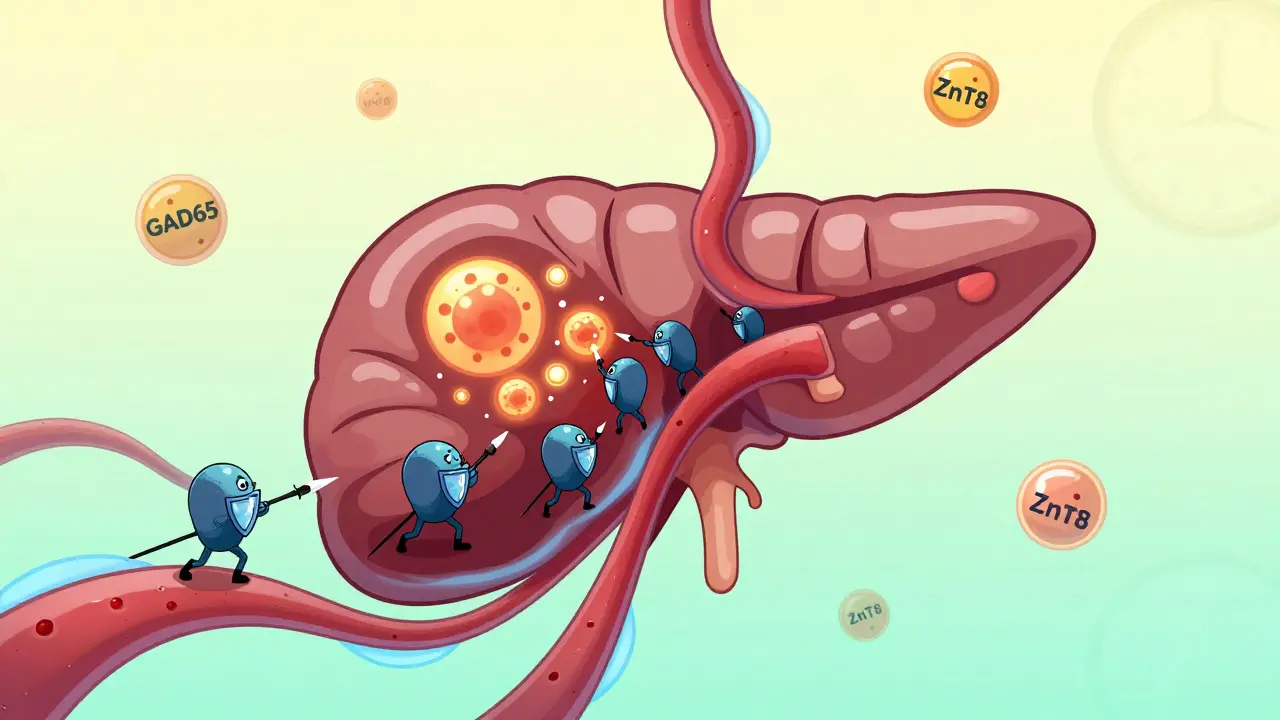
Inside the pancreas, tiny clusters of cells called islets make insulin. In type 1 diabetes, your own immune system sends T-cells to these islets and starts killing the beta cells that produce insulin. This isn’t sudden. It’s a slow, silent war that can start years before symptoms appear. By the time someone feels thirsty, loses weight, or pees constantly, they’ve already lost 80-90% of their insulin-making ability. Blood tests show C-peptide levels below 0.2 nmol/L-proof that the body is barely making any insulin at all.
It’s not random. Genetics play a big role. People with HLA-DR3 or HLA-DR4 gene variants are 20 to 30 times more likely to develop T1D. But genes alone don’t cause it. Something triggers the attack. Viruses like coxsackievirus B show up in the blood months before diagnosis in over half of cases. Diet, gut bacteria, and even early-life antibiotic use might nudge the immune system toward chaos. The beta cells aren’t just passive victims-they may be sending out distress signals that pull the immune system in. That’s why some researchers now say: the beta cell helps start the fight.
Three Stages Before Diagnosis
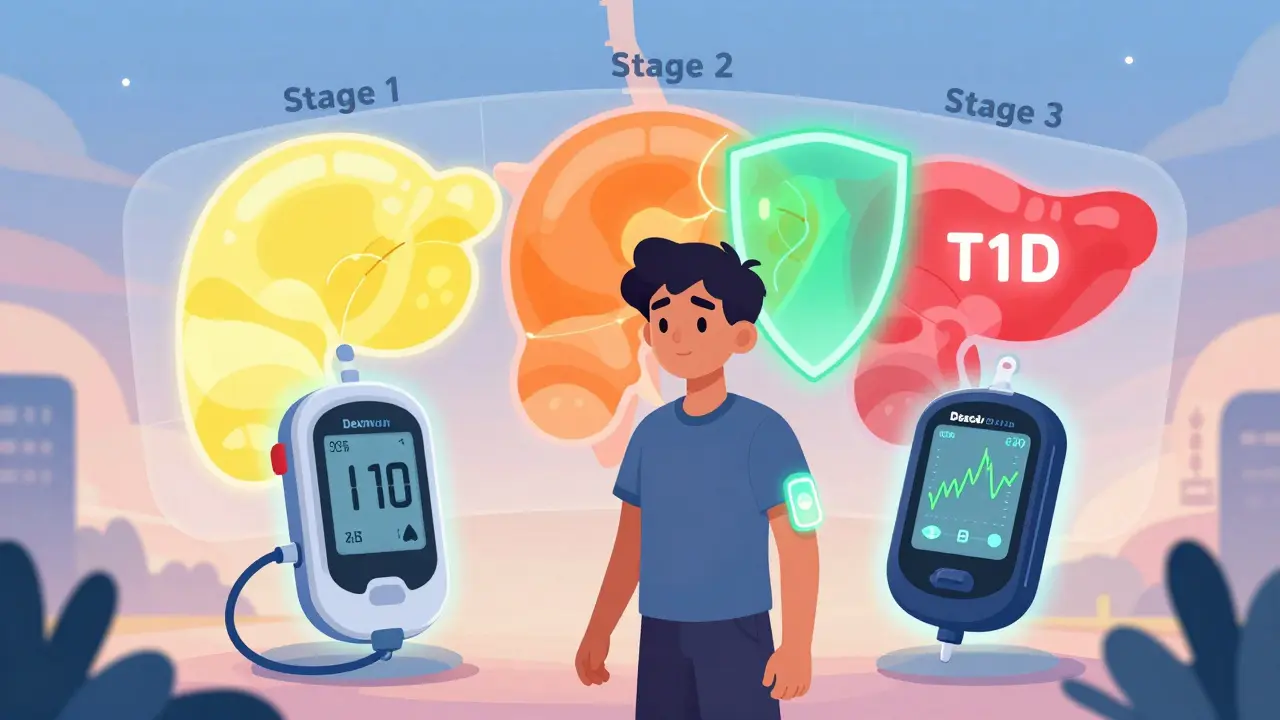
The American Diabetes Association now defines T1D in three clear stages. Stage 1: You have two or more autoantibodies (like GAD65 or ZnT8) in your blood, but your blood sugar is normal. You feel fine. You might not even know you’re at risk. Stage 2: Your blood sugar starts to creep up. You’re still not diabetic by symptoms, but your body is struggling. Stage 3: Symptoms show. You’re diagnosed. This isn’t just medical jargon-it’s a roadmap. If you catch someone in Stage 1 or 2, you might be able to slow or stop the destruction.
That’s where teplizumab (Tzield) comes in. Approved by the FDA in late 2022, it’s the first drug that can delay Type 1 diabetes diagnosis. In clinical trials, it pushed back the onset by over two years in high-risk kids and teens. It doesn’t cure the disease. But it buys time. Time to prepare. Time to learn. Time to avoid diabetic ketoacidosis-the life-threatening emergency that hits 20-30% of kids at diagnosis.
Insulin Isn’t Optional-But How You Use It Matters
You can’t live without insulin. But not all insulin regimens are created equal. The old standard-two shots a day of mixed insulin-is outdated. Today’s gold standard is multiple daily injections (MDI) or insulin pumps. MDI means a long-acting insulin (like glargine U-300) once a day, plus rapid-acting insulin (like aspart or lispro) before every meal. The dose? Usually 0.5 units per kilogram of body weight on day one, split evenly between basal and bolus.
But insulin alone isn’t enough. Blood sugar swings too much without constant monitoring. That’s why continuous glucose monitors (CGMs) like the Dexcom G7 are now essential. They track glucose every five minutes, send alerts when levels dive or spike, and reduce HbA1c by 0.4-0.6% on average. People using CGMs have 40-50% fewer low-blood-sugar episodes. That’s not just convenience-it’s safety.
Even better? Closed-loop systems, or “artificial pancreases.” Devices like Tandem’s Control-IQ automatically adjust insulin based on real-time glucose readings. In 2022, users spent 71-74% of the day in the target range (70-180 mg/dL). Those on traditional pumps? Only 51-55%. For kids and teens, that difference means fewer hospital trips, better sleep, and less anxiety.
The Hidden Pancreas Problem: When Autoimmunity Goes Beyond Beta Cells
Most people with T1D think their pancreas only loses insulin-making ability. But in about 1 in 300 cases, the immune system attacks the whole pancreas-not just the endocrine part, but the exocrine part too. This is autoimmune pancreatitis (AIP), a rare but serious condition. It causes inflammation, pain, bloating, and trouble digesting food. Some patients with T1D develop chronic diarrhea or unexplained weight loss-not because of poor sugar control, but because their pancreas can’t make digestive enzymes.
AIP has two types. Type 1 is linked to high IgG4 levels and responds well to steroids. Type 2 is tied to gut inflammation. When T1D and AIP show up together, treatment gets complicated. Steroids can spike blood sugar. Insulin doses must be adjusted. You need both an endocrinologist and a gastroenterologist working together. The ADA now recommends checking for pancreatic enzyme deficiency in T1D patients with persistent GI symptoms. If it’s there, enzyme replacement therapy can restore digestion and improve nutrient absorption.

New Hope: Beyond Insulin
Research is moving fast. In 2023, Vertex Pharmaceuticals announced that 89% of participants in its first human trial of stem cell-derived islet cells became insulin-independent after 90 days. These lab-grown cells were implanted under the skin and started producing insulin like real beta cells. It’s not a cure yet-patients still need immunosuppressants-but it’s a giant leap.
Other drugs are showing promise too. Verapamil, a blood pressure medication, preserved 30% more insulin production in early-stage T1D patients over 12 months. Abatacept, used for rheumatoid arthritis, slowed beta-cell decline by nearly 60% in recent-onset cases. Even gut health is in focus: people with T1D often lack Faecalibacterium prausnitzii, a gut bacterium that reduces inflammation. Probiotics and fiber-rich diets might help slow progression.
And the cost? Insulin alone costs Americans nearly $10,000 a year. But new therapies-like teplizumab or future stem cell treatments-could cut long-term costs by preventing complications: kidney failure, nerve damage, blindness. Prevention is cheaper than treatment.
What This All Means for Daily Life
If you or someone you know has T1D, here’s what matters now:
- Test for autoantibodies if there’s a family history-even before symptoms.
- Use a CGM. It’s not optional anymore.
- Consider an automated insulin delivery system if you’re still managing with injections.
- Watch for digestive issues. They might not be about diet-they could be about your pancreas.
- Ask about clinical trials. New therapies are opening up fast.
Forget the idea that T1D is just “bad sugar control.” It’s an autoimmune war in the pancreas. And modern management isn’t just about replacing insulin-it’s about protecting what’s left, slowing the attack, and preparing for a future where the body might heal itself again.
Can type 1 diabetes be reversed?
Currently, there is no cure for type 1 diabetes. The immune system continues to attack beta cells even after diagnosis. However, new therapies like teplizumab can delay progression in early stages, and stem cell therapies have restored insulin production in some trial participants. These aren’t cures yet, but they show that stopping or reversing the autoimmune damage is possible in the near future.
Is type 1 diabetes the same as autoimmune pancreatitis?
No. Type 1 diabetes targets the insulin-producing beta cells in the pancreas (endocrine function). Autoimmune pancreatitis attacks the enzyme-producing cells (exocrine function), causing digestive problems. They are separate diseases, but in rare cases (about 0.3% of T1D patients), both occur together, suggesting a broader autoimmune disorder affecting the whole pancreas.
Why do some adults get misdiagnosed with type 2 diabetes?
About 12% of adults with type 1 diabetes are initially misdiagnosed as having type 2-especially if they’re overweight or older. Doctors assume insulin resistance, not autoimmunity. But if someone has autoantibodies, low C-peptide, and no signs of metabolic syndrome, it’s likely type 1. Testing for antibodies and C-peptide can prevent this error.
Do I need to avoid carbs if I have type 1 diabetes?
No. You don’t need to avoid carbs. You need to match insulin to the carbs you eat. Modern insulin regimens and CGMs make it possible to eat a wide variety of foods. The goal isn’t restriction-it’s balance. Work with a dietitian to learn how to count carbs, adjust insulin, and keep blood sugar steady without eliminating entire food groups.
Can lifestyle changes stop type 1 diabetes from getting worse?
Lifestyle changes alone can’t stop the autoimmune attack. But they can help reduce complications. Eating well, staying active, and avoiding smoking improve heart and kidney health. Managing stress and sleep helps blood sugar control. Some research suggests a healthy gut microbiome may slow beta-cell loss. While they won’t cure T1D, healthy habits make living with it easier and safer.

Comments (8)